How Fuel Cells Transform Hydrogen into Clean Energy
Introduction
As energy systems seek to reduce emissions while maintaining reliability and performance, fuel cells are gaining visibility. By converting hydrogen into electricity without combustion, they offer a credible alternative to conventional thermal technologies in transportation, industry, and stationary energy production.
This electrochemical approach relies on key components and distinct operating principles, which explain the growing interest in this technology.
What is a Fuel Cell?
A fuel cell represents a revolution in energy production by directly converting chemical energy into electricity without an intermediate combustion step. According to the U.S. Department of Energy, this technology uses the chemical energy of hydrogen or other fuels to produce electricity cleanly and efficiently, generating only electricity, water, and heat when hydrogen is used as fuel [1].
A Fundamental Difference from Batteries
Unlike batteries that store chemical energy and must be regularly recharged, fuel cells generate energy continuously. They function like batteries, but do not discharge and do not need to be recharged as long as they are supplied with fuel [1]. This characteristic makes them particularly suited for applications requiring sustained energy production.
The Electrochemical Principle
The process relies on a controlled chemical reaction that separates hydrogen atoms into protons and electrons. This separation creates an electrical flow while producing only water as a by-product. The absence of combustion eliminates the pollutant emissions typical of conventional technologies.
Remarkable Energy Efficiency
Fuel cells can operate with efficiencies exceeding 60% [1], which represents a considerable advantage. To put this figure in perspective, a typical car's gasoline engine converts only about 20% of the fuel's energy into motion under normal driving conditions [2]. This exceptional efficiency is explained by the elimination of intermediate energy conversion steps present in combustion systems.
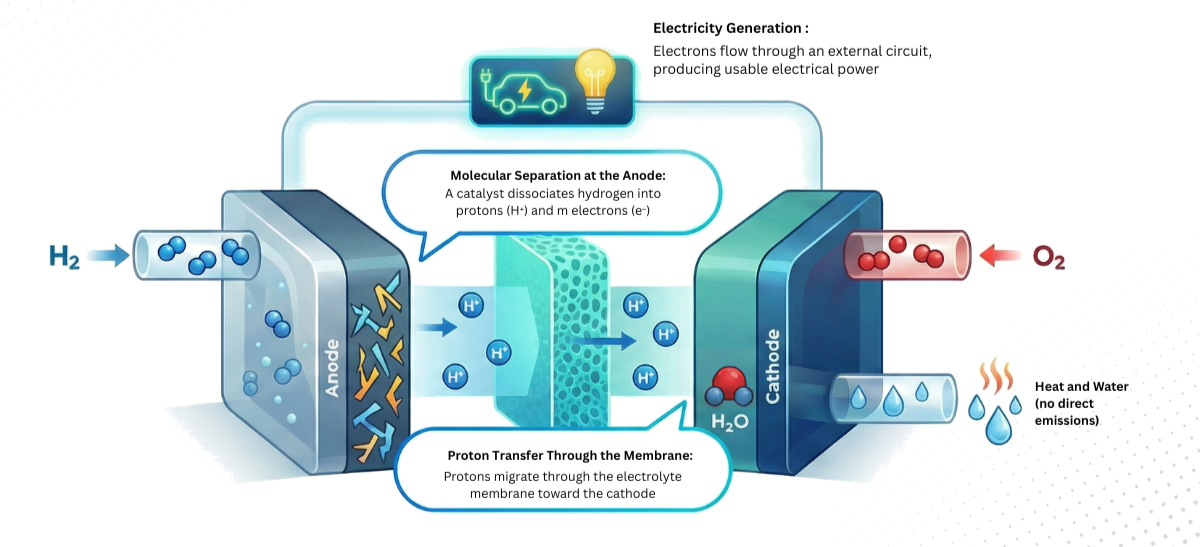
How a Fuel Cell Works
Understanding the internal mechanism of a fuel cell helps grasp why this technology is so efficient and clean. The process unfolds in four distinct but interconnected steps.
| Step | Step Name | What Happens | Key Result |
|---|---|---|---|
| 1 | Supply | Hydrogen is injected at the anode (–) and oxygen from the air is directed to the cathode (+). Both reactants are kept separate. | Preparation of reactants |
| 2 | Molecular Separation | A catalyst at the anode breaks down hydrogen molecules into protons (H⁺) and electrons (e⁻). | Creation of electrical charges |
| 3 | Electricity Production | Electrons cannot pass through the electrolytic membrane and therefore flow through an external circuit. This movement of electrons through the circuit creates a usable electrical current to power devices, vehicles, or buildings. | Generation of an electrical current |
| 4 | Recombination and Water Production | While electrons flow through the external circuit, protons migrate through the electrolytic membrane to the cathode. There, they combine with oxygen from the air and electrons arriving from the external circuit to produce water and heat. | Water + heat as by-products |
A Major Thermodynamic Advantage
Fuel cells can convert the chemical energy of fuel directly into electrical energy with efficiencies that can exceed 60% [1]. In comparison, a conventional car's gasoline engine has an efficiency of less than 20% under normal driving conditions [2].
This superiority is explained by a fundamental thermodynamic principle. Combustion engines must first convert fuel into heat, then that heat into mechanical energy, and finally that mechanical energy into electricity. Each conversion entails energy losses. Fuel cells, by operating directly from electrochemical reactions, avoid these intermediate steps and their associated losses [3].
Continuous Energy Production
As long as hydrogen and oxygen are supplied to the cell, it produces electricity uninterruptedly. This continuous operating capability fundamentally distinguishes it from a battery, which gradually accumulates chemical by-products and must be periodically recharged.
The Architecture of a Fuel Cell: Essential Components
Each component of a fuel cell plays a precise role in converting hydrogen into electricity. Together, they form an electrochemical system optimized to deliver high efficiency and clean energy production.
The Proton Exchange Membrane (PEM): The polymer electrolyte membrane forms the heart of the fuel cell. This very thin material, comparable to a plastic film, allows only positively charged ions to pass through while blocking electrons. This property forces electrons to flow through an external circuit, producing electricity. For transportation applications, the membrane is extremely thin and can measure less than 20 microns [4].
The Catalyst Layers: Located on either side of the membrane, the catalyst layers facilitate electrochemical reactions. They are generally composed of nanometric platinum particles deposited on a carbon support. At the anode, the catalyst separates hydrogen into protons and electrons, while at the cathode, it enables the reaction between oxygen, protons, and electrons arriving from the external circuit to form water [4].
Progressive Reduction of Platinum Use: Since platinum is a rare and costly metal, research efforts aim to reduce the quantity used. Recent catalysts use approximately one quarter of the platinum quantity of previous technologies, without notable loss of performance [5]. In parallel, catalysts without platinum group metals, notably based on iron-nitrogen-carbon, have achieved power densities of 0.85 W/cm² and stability exceeding 1,000 hours, improving the economic accessibility of the technology [6].
Gas Diffusion Layers (GDL): Gas diffusion layers ensure a uniform distribution of hydrogen and oxygen toward the catalytic sites, while facilitating the evacuation of water produced by the reaction. Effective water management is essential to prevent flooding of catalytic sites and maintain fuel cell performance.
Bipolar Plates and Cell Stacking: Each membrane-electrode assembly produces less than 1 volt under normal operating conditions [4]. To reach usable voltages, several cells are stacked in series. Bipolar plates handle current collection and gas distribution, allowing power and voltage to be adapted to the needs of transportation, buildings, or industrial applications.
The Different Types of Fuel Cells
Depending on the operating temperature and intended application, different types of fuel cells have been developed, each with specific advantages.
PEM Fuel Cells (Polymer Electrolyte Membrane)
PEM fuel cells operate at relatively low temperatures, around 80°C [7]. This moderate temperature confers several practical advantages: they can start up quickly, requiring less warm-up time than other types. Furthermore, low-temperature operation causes less wear on system components, which considerably improves durability.
These characteristics make PEM fuel cells ideal for automotive transportation, where frequent starts and long-term reliability are crucial. They are also suitable for portable applications and small-scale residential systems.
SOFC Fuel Cells (Solid Oxide)
At the opposite end of the thermal spectrum are solid oxide fuel cells, which operate at very high temperatures that can reach 1,000°C [7]. Despite this thermal constraint, they offer a remarkable efficiency of around 60% for converting fuel into electricity.
The major advantage of SOFCs lies in their cogeneration capability. In applications designed to capture and use the residual heat from the system, overall fuel utilization efficiencies could exceed 85% [7]. This exceptional efficiency makes them particularly attractive for large-scale stationary applications, such as decentralized power plants or industrial facilities simultaneously requiring electricity and heat.
MCFC Fuel Cells (Molten Carbonate)
Molten carbonate fuel cells operate at intermediate temperatures, around 650°C. When coupled with a turbine, they can achieve efficiencies approaching 65%, which is considerably higher than the 37–42% efficiency of a phosphoric acid fuel cell plant [7].
As with SOFCs, when residual heat is captured and used in cogeneration systems, overall fuel efficiencies can exceed 85%. These performances make them prime candidates for industrial electricity production and large stationary installations.
Comparison Table
| Cell type | Operating temperature | Main applications | Electrical efficiency |
|---|---|---|---|
| PEM | 80 °C | Transport, portable applications | 40–60 % |
| SOFC | 1000 °C | Stationary installations | 60 % (up to 85 % with cogeneration) |
| MCFC | 650 °C | Industrial power generation | Up to 65 % |
Environmental and Energy Advantages
Fuel cells offer significant advantages for the global energy transition, both in terms of efficiency and environmental impact.
Zero Emissions at Point of Use
When operating on hydrogen, fuel cells produce only water and heat at the point of use. In the absence of combustion, they emit no greenhouse gases, nitrogen oxides, or fine particles during operation. This characteristic is a major advantage for air quality, particularly in densely populated urban areas.
Substantial Reduction in Greenhouse Gas Emissions
Recent data demonstrates that hydrogen fuel cell vehicles consume approximately 29 to 66% less energy and cause approximately 31 to 80% fewer greenhouse gas emissions than conventional vehicles [8]. This massive reduction in emissions represents an important lever for achieving decarbonization objectives in the transportation sector.
Silent Operation
An often underestimated advantage of fuel cells is their silent operation. Fuel cells have no moving parts, making them potentially far more reliable than combustion engines, while being considerably quieter [3].
Multi-Sector Applications
The versatility of fuel cells is a major asset. They can be used across a wide range of applications, providing energy for multiple sectors: transportation (light vehicles, buses, trucks, trains, ships), industrial, commercial, and residential buildings, as well as long-term energy storage for the electrical grid in reversible systems [1]. This flexibility allows the technology to be adapted to the specific needs of each application.
White Hydrogen: An Emerging Natural Supply Source
While fuel cells enable hydrogen to be converted into electricity efficiently and cleanly, their large-scale deployment is closely tied to hydrogen supply sources. As the fuel cell sector matures, increasing attention is being paid to the origin of the hydrogen used and its overall environmental footprint.
In this context, the emergence of white hydrogen — a form of hydrogen naturally present in the Earth's subsurface — represents a development closely linked to the rise of fuel cell technologies. This potential supply source could, in time, complement existing supply chains and help structure a more sustainable hydrogen ecosystem.
An Emerging Geological Resource
Natural hydrogen, also called white hydrogen, represents a long-neglected geological energy source that could reinforce the low-carbon character of the global energy portfolio [9]. Unlike hydrogen produced industrially through methane reforming or electrolysis, it is naturally generated in the Earth's subsurface, making it a subject of growing interest for players in the hydrogen sector.
Conclusion
Fuel cells represent a mature and promising technology for the energy transition. With efficiencies reaching 60% — approximately three times that of a conventional combustion engine — they offer an effective solution for converting hydrogen into electricity without pollutant emissions at the point of use. The global market is experiencing exceptional growth of 20% per year, reflecting the growing interest of industries and governments in this technology.
The architecture of fuel cells continues to evolve, notably with the progressive reduction in platinum use and the development of catalysts without precious metals. These advances make the technology increasingly economically accessible and pave the way for large-scale deployment.
Alongside technological advances in fuel cells, growing interest in white hydrogen highlights the importance of supply sources in the development of the hydrogen sector. As a natural geological resource, it is distinguished by a particularly low carbon footprint compared to conventional supply chains, while suggesting the potential for significant volumes. These characteristics could, in time, contribute to supporting the large-scale deployment of fuel cell technologies.
Fuel cells are not a distant future technology, but a current reality already integrated into transportation, industry, and stationary energy production. Coupled with white hydrogen, they could significantly accelerate the decarbonization of our economy and contribute decisively to achieving global climate objectives.
To learn more about energy innovations and Squatex's projects in the field of natural hydrogen and renewable energies, follow us on LinkedIn.
References
[1] U.S. Department of Energy. "Fuel Cells." Office of Energy Efficiency & Renewable Energy, https://www.energy.gov/eere/fuelcells/fuel-cells.
[2] U.S. Department of Energy. "Fuel Cells Fact Sheet." Office of Energy Efficiency & Renewable Energy, November 2015, https://www.energy.gov/sites/prod/files/2015/11/f27/fcto_fuel_cells_fact_sheet.pdf.
[3] Environmental and Energy Study Institute. "Fact Sheet: Fuel Cells." EESI, https://www.eesi.org/papers/view/fact-sheet-fuel-cells.
[4] U.S. Department of Energy. "Parts of a Fuel Cell." Office of Energy Efficiency & Renewable Energy, https://www.energy.gov/eere/fuelcells/parts-fuel-cell.
[5] Argonne National Laboratory. "Argonne scientists maximize the effectiveness of platinum in fuel cells." Argonne News, December 2024, https://www.anl.gov/article/argonne-scientists-maximize-the-effectiveness-of-platinum-in-fuel-cells.
[6] International Journal of Low-Carbon Technologies. "Enhancement in fuel cells: PGM-free catalysts, nanostructured supports, and advanced membrane technology toward low-carbon emission." Oxford Academic, January 24, 2025, https://academic.oup.com/ijlct/article/doi/10.1093/ijlct/ctaf008/8006312.
[7] U.S. Department of Energy. "Types of Fuel Cells." Office of Energy Efficiency & Renewable Energy, https://www.energy.gov/eere/fuelcells/types-fuel-cells.
[8] Fastech. "Hydrogen Fuel Cell Efficiency." Fastech Blog, https://www.fastechus.com/blog/hydrogen-fuel-cell-efficiency/.
[9] U.S. Geological Survey. "Potential for Geologic Hydrogen: The Next Generation Energy." USGS Featured Story, https://www.usgs.gov/news/featured-story/potential-geologic-hydrogen-next-generation-energy.